Citizen Health × CharacterQuilt
Four campaigns. One afternoon.
Designed by AI, finished by humans.
A complete go-to-market kit built for Citizen Health Life Sciences — landing pages, display ads, decks, one-pagers, ebook covers, and nurture sequences across four target angles.
↓
Pick a campaign
Four campaign angles for one recontactable rare-disease cohort.
Citizen Health Life Sciences sells the same platform — regulatory-grade real-world data + recontactable patient registries — to four very different audiences. We built the kit for each.
Novartis — gene-therapy programs need a cohort that's already in place.
We pitched Novartis Rare Disease teams the angle that compresses pre-IND, pivotal, and post-market evidence onto one longitudinal cohort. Recontactable, FDA-accepted, ready before they file.
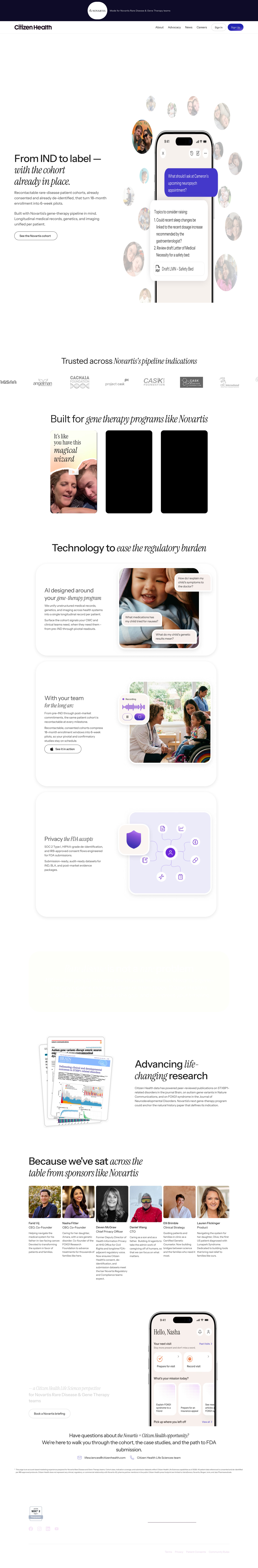 Landing Page
Landing Page
Personalized Landing Page
From IND to label, with the cohort already in place.
2 Master Ads
Display Ads — Master
Dark + light angles, 1024×1024
12 Resizes
Platform Resizes
6 sizes × 2 master angles
 Ebook
Ebook
Ebook Cover · US Letter
The Novartis playbook for gene-therapy RWE
 8-Slide Deck
8-Slide Deck
Sales Deck
Cover · context · pain · solution · proof · ROI · integration · CTA
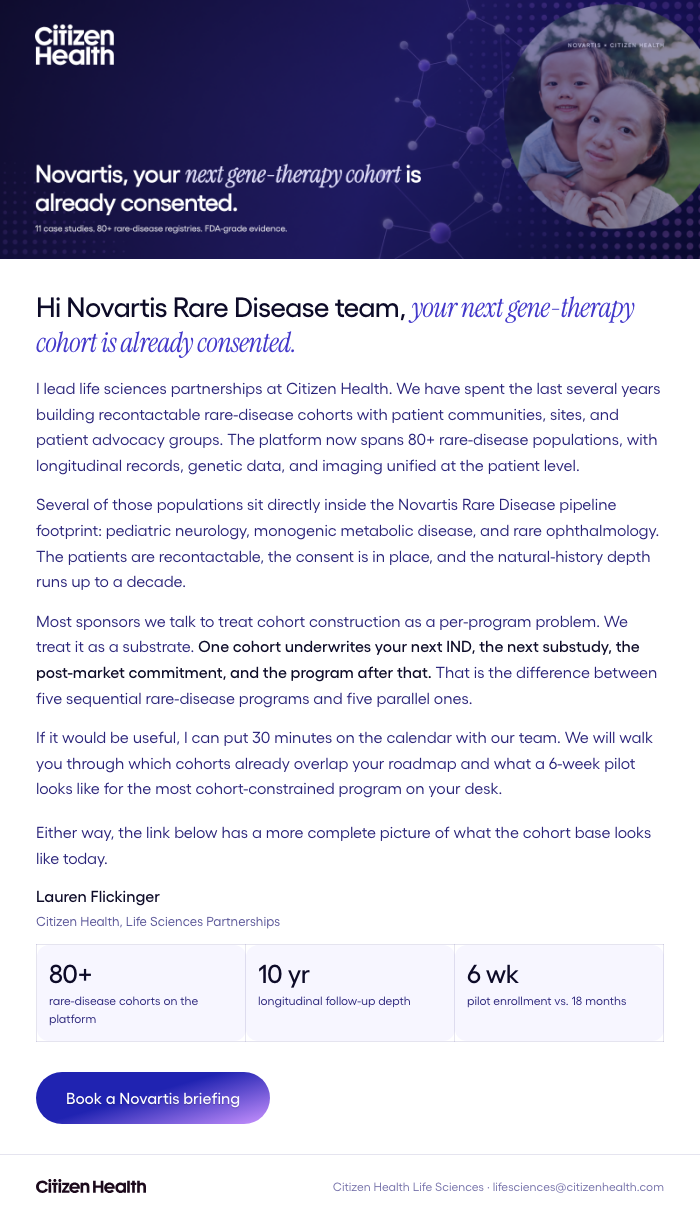 3-Email Sequence
3-Email Sequence
Nurture Emails
Intro → Proof → Closing
Biogen — neurology label expansions need decade-long longitudinal data.
From SMA Spinraza to Friedreich's Skyclarys to Alzheimer's Leqembi — every Biogen label depends on RWE that only a recontactable patient registry can produce. We pitched Biogen Neurology the cohort infrastructure.
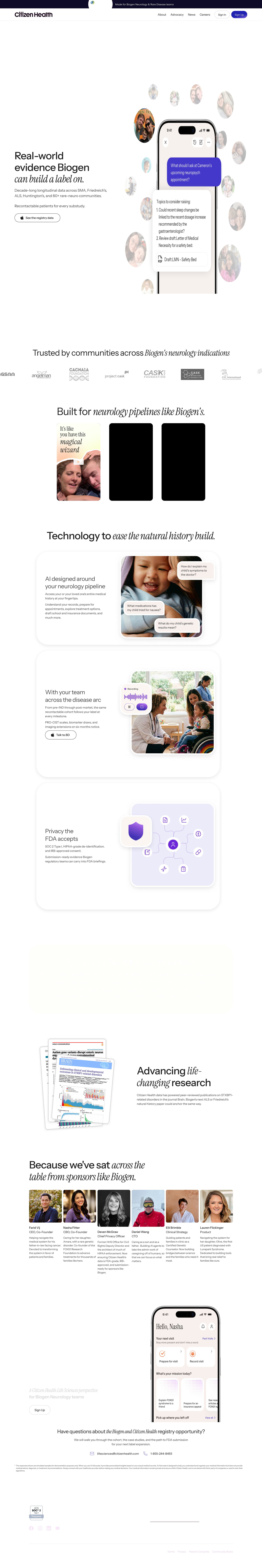 Landing Page
Landing Page
Personalized Landing Page
Real-world evidence Biogen can build a label on.
2 Master Ads
Display Ads — Master
Dark + light angles, 1024×1024
12 Resizes
Platform Resizes
6 sizes × 2 master angles
 Ebook
Ebook
Ebook Cover · US Letter
The neurology RWE playbook for the Biogen pipeline
 8-Slide Deck
8-Slide Deck
Sales Deck
Cover · context · pain · solution · proof · ROI · integration · CTA
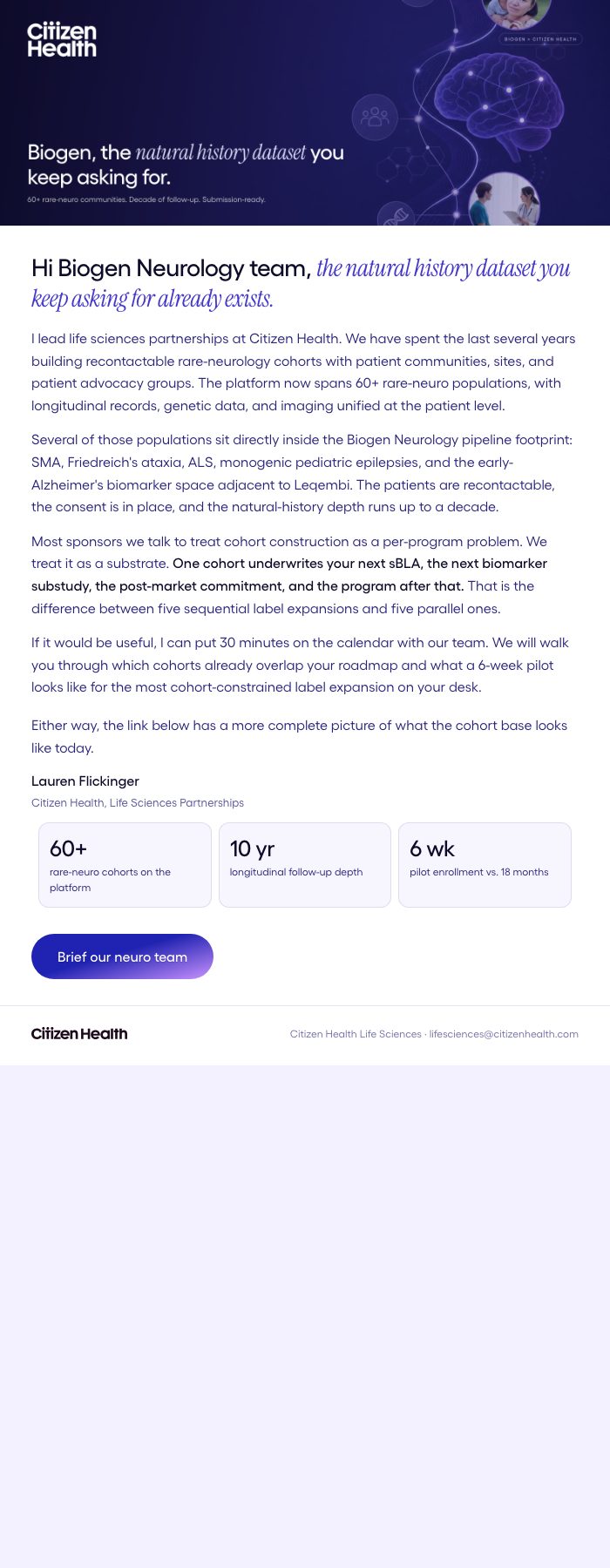 3-Email Sequence
3-Email Sequence
Nurture Emails
Intro → Proof → Closing
Academic Medical Centers — investigator-led research, without the registry build.
UCSF, St. Jude, CHOP, Boston Children's, Emory and Mass General PIs need recontactable cohorts in 6 weeks, not 18 months. Citizen Health data already powered a Brain journal paper on STXBP1 — yours next.
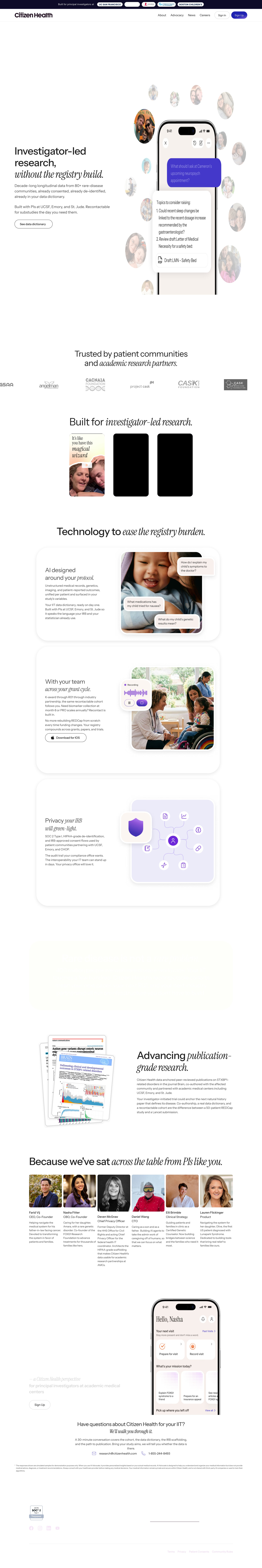 Landing Page
Landing Page
Personalized Landing Page
Investigator-led research, without the registry build.
2 Master Ads
Display Ads — Master
Dark + light angles, 1024×1024
12 Resizes
Platform Resizes
6 sizes × 2 master angles
 Ebook
Ebook
Ebook Cover · US Letter
The investigator's guide to rare-disease real-world data
 8-Slide Deck
8-Slide Deck
Sales Deck
Cover · context · pain · solution · proof · ROI · integration · CTA
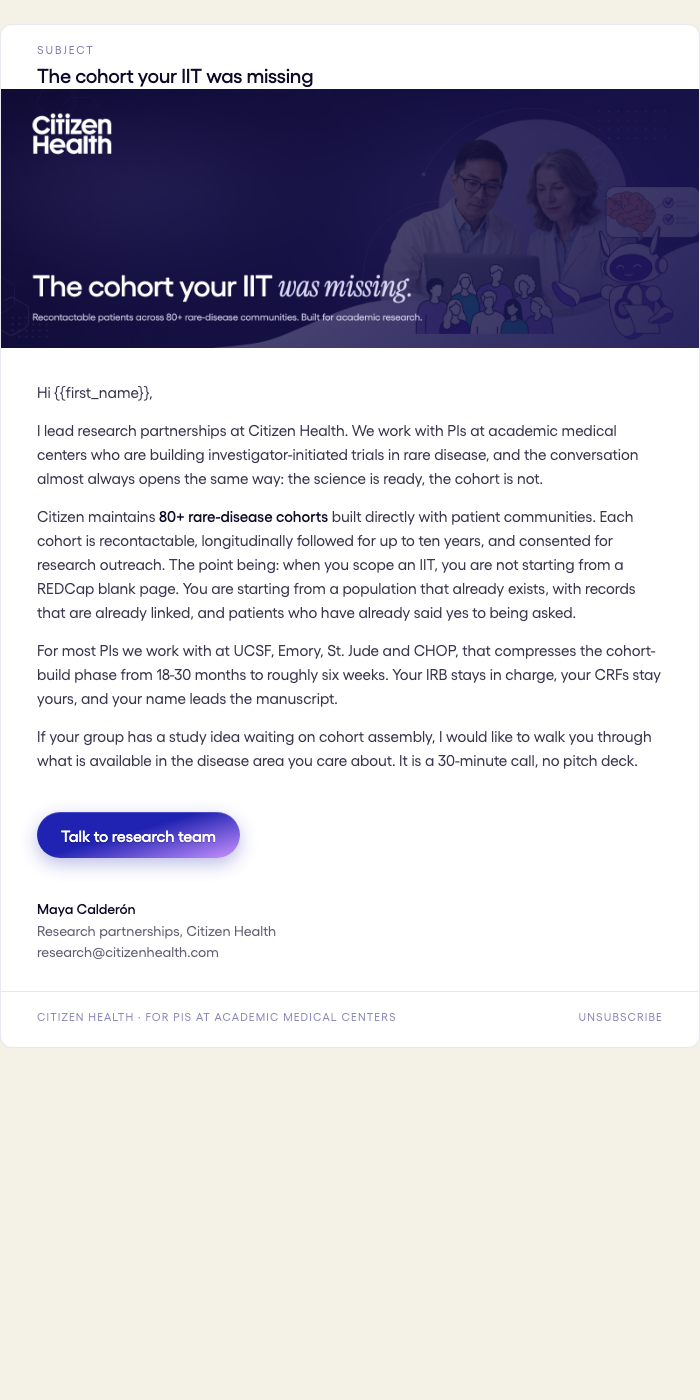 3-Email Sequence
3-Email Sequence
Nurture Emails
Intro → Proof → Closing
Patient advocacy organizations — your foundation deserves a real registry, not a spreadsheet.
80+ rare-disease foundations already run their patient community on Citizen Health. We pitched the next batch — NORD, Global Genes, EveryLife, and the founders building the next CACNA1A or Cohen Syndrome Foundation.
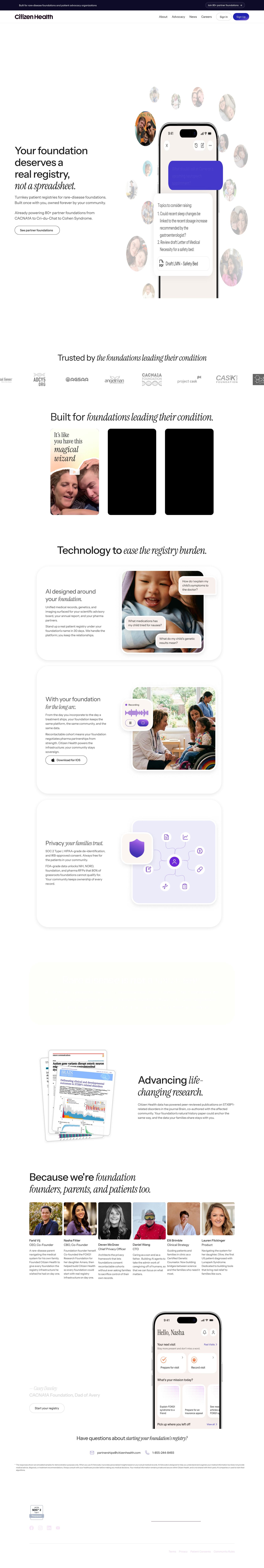 Landing Page
Landing Page
Personalized Landing Page
Your foundation deserves a real registry, not a spreadsheet.
2 Master Ads
Display Ads — Master
Dark + light angles, 1024×1024
12 Resizes
Platform Resizes
6 sizes × 2 master angles
 Ebook
Ebook
Ebook Cover · US Letter
The foundation's playbook for patient registry success
 8-Slide Deck
8-Slide Deck
Sales Deck
Cover · context · pain · solution · proof · ROI · integration · CTA
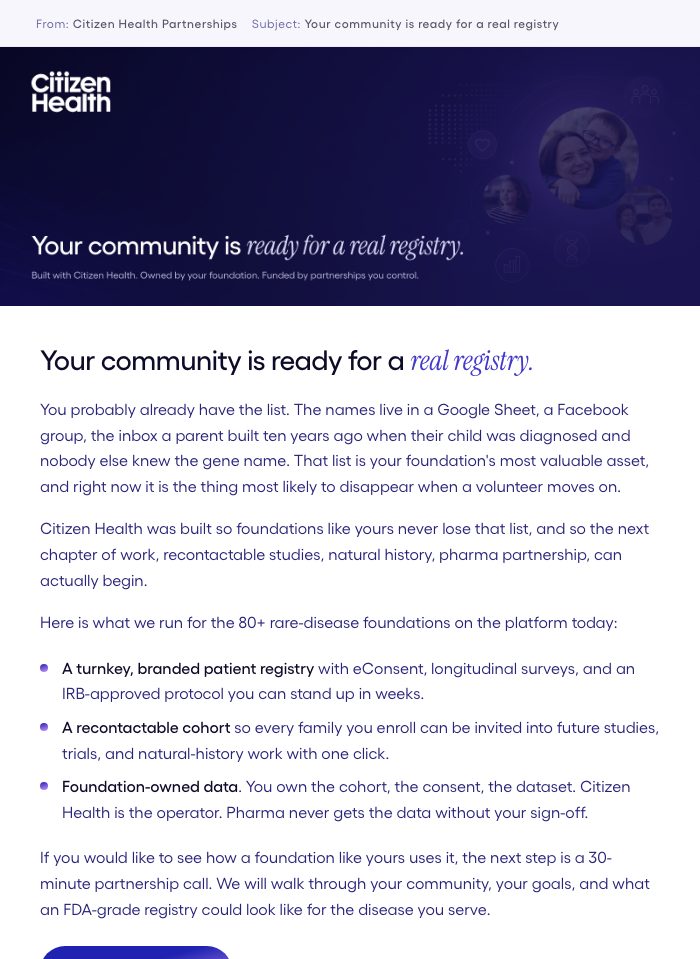 3-Email Sequence
3-Email Sequence
Nurture Emails
Intro → Proof → Closing



































